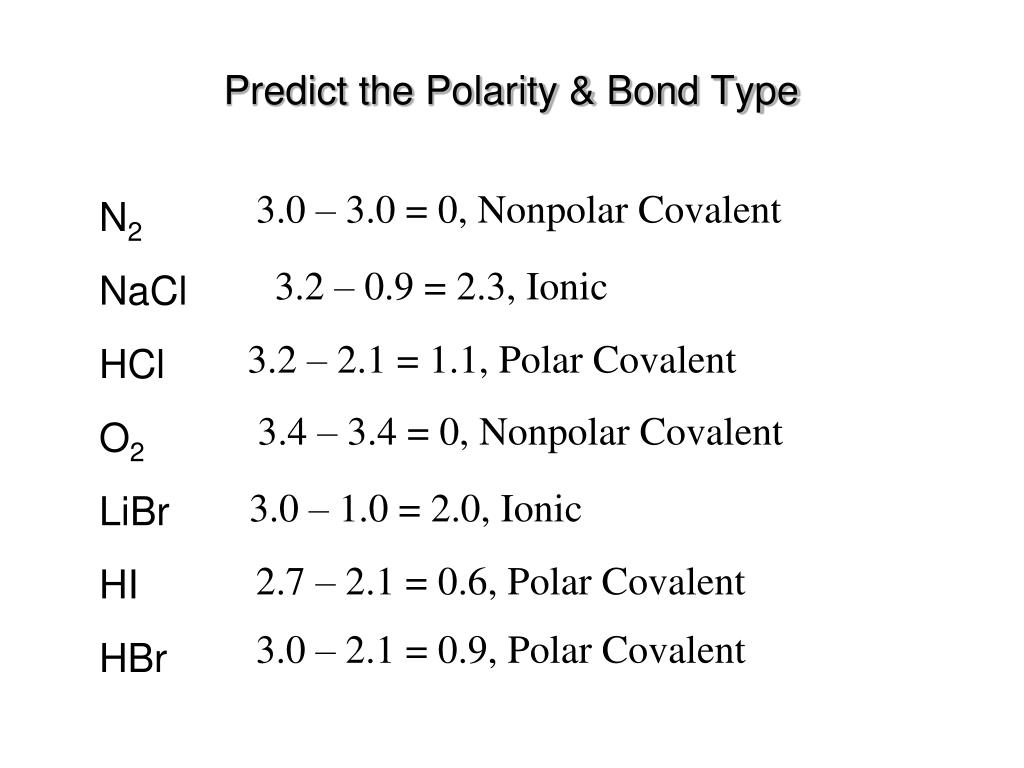
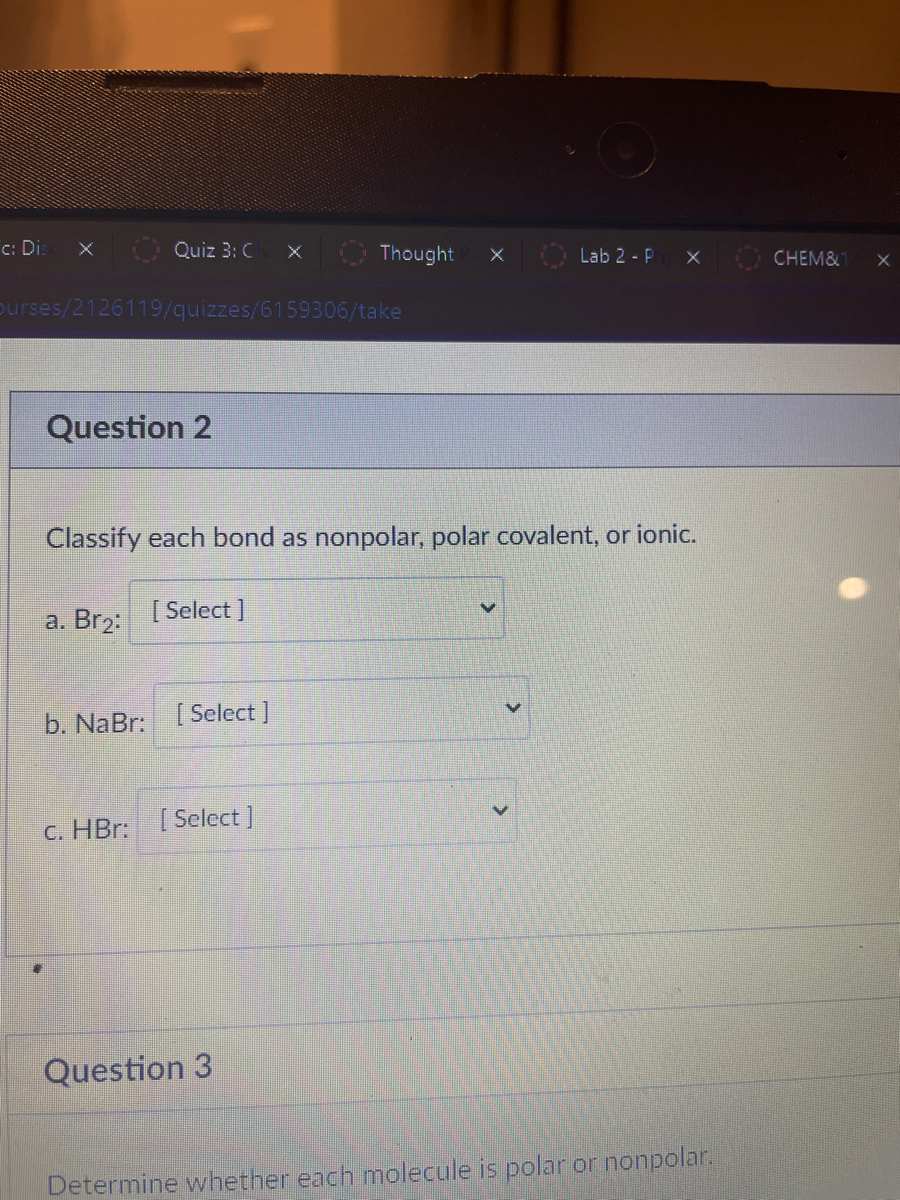
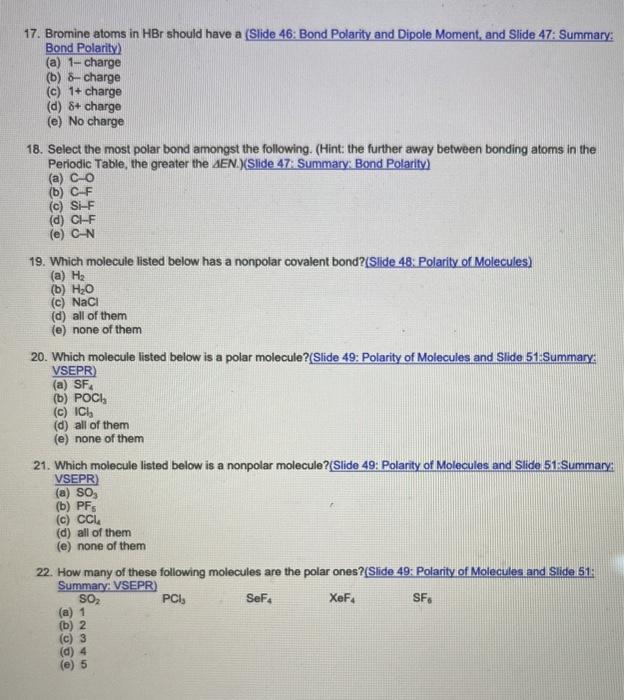
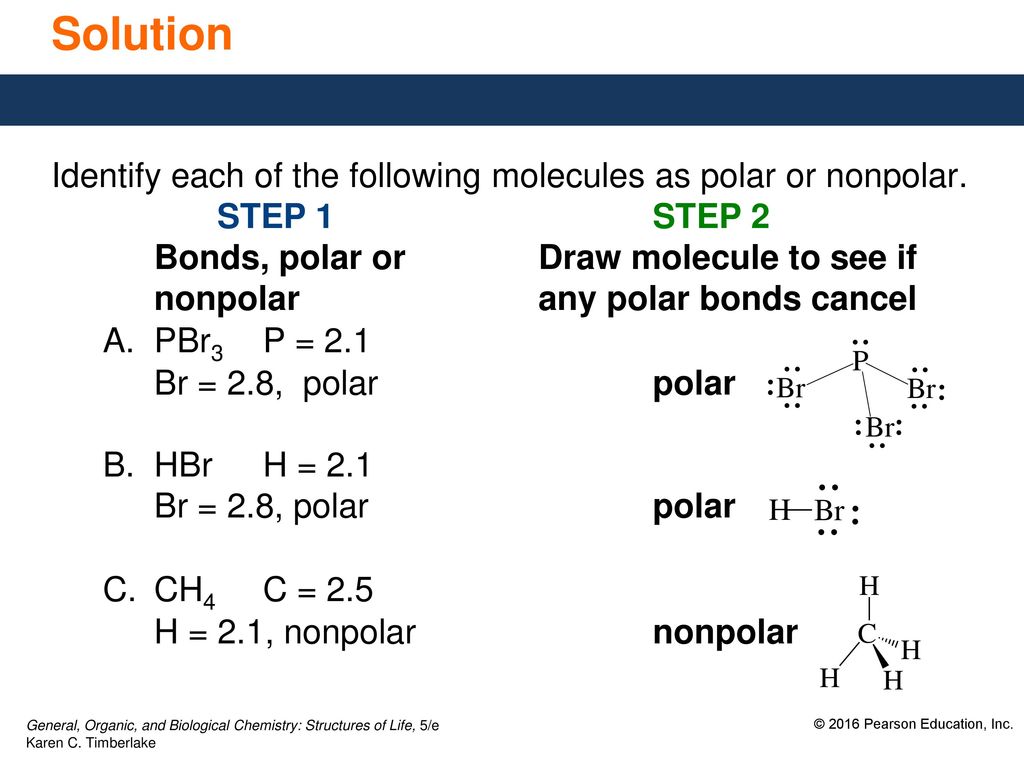
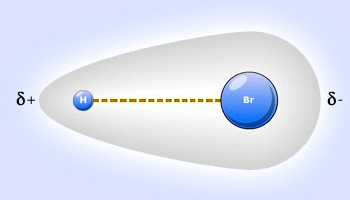
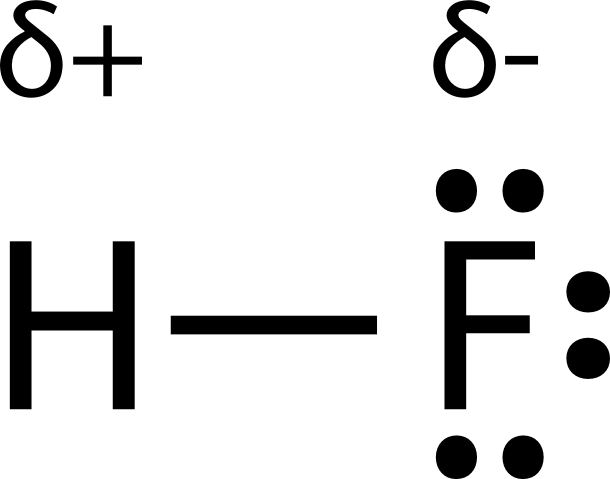SOLVED: Text: Identify the polarity of these molecules and classify them as polar or nonpolar. Drag each item to the appropriate bin. View Available Hint(s) HBc BBeg CBr4 CH3NH2 H2O Polar Nonpolar

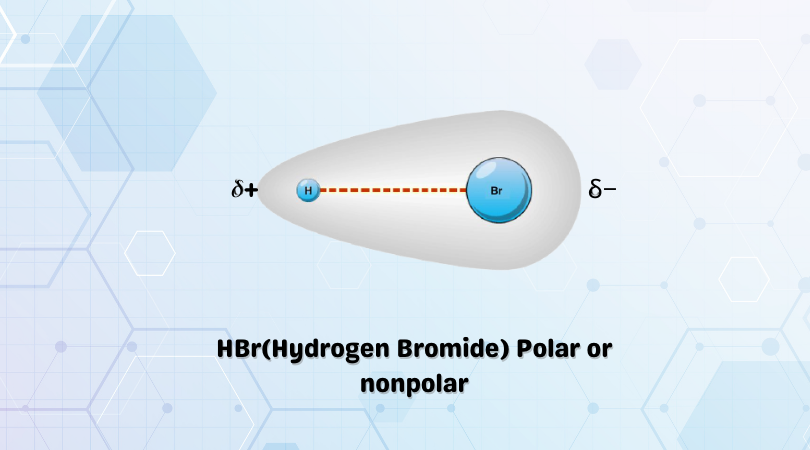
Hydrogen bromide gas, polar covalent compound, is very soluble in water. Describe what happens, referring to intermolecular forces, as the hydrogen bromide dissolves. | Homework.Study.com